|
3/9/2024 0 Comments Electron geometry chart of pf3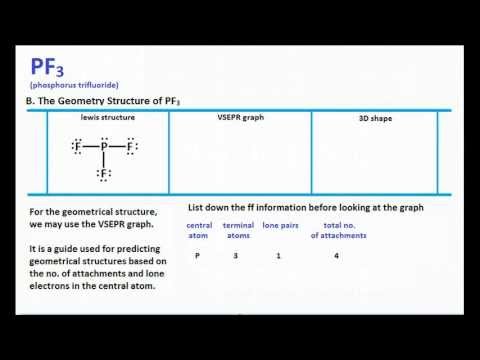
Although the bond angle should be 109.5 degrees for trigonal pyramidal molecular geometry, it decreases to 107 degrees due to the lone pair on the nitrogen atom. This pair exerts repulsive forces on the bonding pairs of electrons. The shape is distorted because of the lone pairs of electrons. It has a molecular geometry of trigonal pyramidal which also looks like a distorted tetrahedral structure. 
There are three single bonds and one lone pair of electrons in the NH3 molecule. Thus, Ammonia or NH3 has sp3 hybridization. When it shares the electrons with Hydrogen atoms, one s-orbital and three p-orbitals hybridize and overlap with s orbitals of a Hydrogen atom to form sp3 hybridization. The Nitrogen atom has the electronic configuration of 1s2 2s2 2px1 2py1 2pz1. All the Hydrogen atoms are arranged symmetrically around the Nitrogen atom which forms the base, and the two nonbonding electrons form the tip which makes the molecular geometry of NH3 trigonal pyramidal. NH3 Molecular GeometryĪmmonia has a tetrahedral molecular geometry. Thus there are three single bonds formed between Nitrogen and Hydrogen atoms, and there is one pair of nonbonding electrons on the nitrogen atom. Nitrogen will share three of its valence electrons for forming a stable structure. Place all the Hydrogen atoms around the Nitrogen atom and the valence electrons of both the atoms like this.Įach Hydrogen atom only needs one electron to become stable, as it is an exception to the octet rule. Hydrogen atoms never take the central position, so we will place the Nitrogen atom in the center. Now that we know the valence electrons for the molecule, we can predict its Lewis structure. Here is the step-by-step procedure to understand the Lewis structure of NH3. The electrons that form bonds are called bonding pair of electrons, whereas the ones that do not form any bonds are called nonbonding pairs of electrons or lone pairs of electrons.ĭots are used to show the valence electrons, whereas the lines represent bonds in the structure. It is a pictorial representation of the arrangement of valence electrons around the individual atoms in the molecule. It often serves as a ligand in complex metal compounds, binding to metal atoms through its lone pair of electrons. The Lewis structure of a molecule helps understand the electron geometry, molecular geometry, polarity, and other such properties with ease. PF 3 is a Lewis acid, meaning it can accept electron pairs. Hydrogen – 1 electron, but as there are 3 Hydrogen atoms we will multiply it by 3, there are three valence electrons of all Hydrogen atoms.Īmmonia or NH3 has a total of 8 valence electrons. To get the total number of valence electrons, we will add up the valence electrons for both these atoms. In contrast, Hydrogen is a group 1 element and only has 1 valence electron in its outer shell. Nitrogen is a group 15 element and has five electrons in its outer shell. NH3 Bond angles Valence electrons of NH3 ( Ammonia ).Arsenin, Solutions of Ill-Posed Problems (Nauka, Moscow, 1974 Winston, Washington, D.C., 1977). Absolute partial electron ionisation cross sections, and precursor-specific partial electron ionisation cross sections, for the formation of cations from phosphorus trifluoride (PF 3) are reported over the electron energy range 50200 eV. Kreyszig, Advanced Engineering Mathematics, 8th ed. Kostomarov, Numerical Simulation of Plasmas (Nauka, Moscow, 1982 Springer, New York, 1986). 67, 843 (2015).įreeGS: Free boundary Grad-Shafranov solver. GitHub, D3DEnergetic/FIDASIM: A Neutral Beam and Fastion Diagnostic Modeling Suite. Hooper, Report UCRL-ID126284 (Lawrence Livermore National Laboratory, Livermore, CA, 1997). Yushmanov, Report No. IPP-5/98 (Max-Planck-Institut für Plasmaphysik, Garching, 2002). by B. Coppi (Commission of the European Communities, Brussels, 1981), Vol. Hawryluk, in Physics of Plasmas Close to Thermonuclear Conditions: Proceedings of the Course Held in Varenna, Italy 27 August−8 September 1979, Ed. 31: Theoretical and Experimental Aspects of Controlled Nuclear Fusion (UN, Geneva, 1958), p. Rubin, in Proceedings of the 2nd International Conference on the Peaceful Uses of Atomics E-nergy held in Geneva, 1 September−13 September 1958, Vol.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
